January 10, 2014
Working with colleagues in India and England, we recently published the paper, "Association between being employed in a smoke-free workplace and living in a smoke-free home: Evidence from 15 low and middle income countries," in Preventive Medicine. We found that, on average, people covered by legislation requiring smokefree workplaces and public places are 60% more likely to implement voluntary policies making their homes smokefree.
Here is the abstract:
Objective:To assess whether being employed in a smoke-free workplace is associated with living in a smoke-free home in 15 low and middle income countries (LMICs).
Methods: Country-specific individual level analyses of cross-sectional Global Adult Tobacco Survey data (2008–2011) from 15 LMICs was conducted using multiple logistic regression. The dependent variable was living in a smoke-free home; the independent variable was being employed in a smoke-free workplace. Analyses were adjusted for age, gender, residence, region, education, occupation, current smoking, current smokeless tobacco use and number of household members. Individual country results were combined in a random effects meta-analysis.
January 10, 2014
Eight years ago, in 2006, US District Court Judge Gladys Kessler convicted the big cigarette companies and their trade and scientific groups of forming an illegal racketeering "enterprise" to defraud the American people and, among other things, ordered the cigarette companies to publish "corrective statements" telling the public that they had lied about the dangers of smoking, secondhand smoke, and nicotine addiction. (Judge Kessler also prohibited them from challenging the evidence that these statements are true, which is why the companies no longer do so.)
Judge Kessler ordered the defendant tobacco companies to work with the Department of Justice and the public health intervenors to determine just how the corrective statements would be made on the internet, in print, and on television and radio. These negotiations have now concluded and a draft ruling submitted to the Court.
January 8, 2014
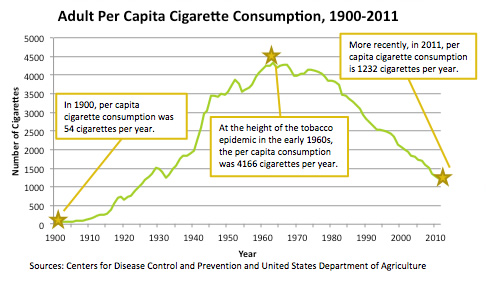 I have been getting many press calls for my assessment of the importance of the 1964 Surgeon General's report. There is no question that the report was a landmark event that provided the weight of government behind the conclusion, already reached by the scientific community 10 years earlier, that smoking caused lung cancer. The issuance of the report and the attendant public discussion did seem to stop the growth in per capita cigarette consumption (graph).
I have been getting many press calls for my assessment of the importance of the 1964 Surgeon General's report. There is no question that the report was a landmark event that provided the weight of government behind the conclusion, already reached by the scientific community 10 years earlier, that smoking caused lung cancer. The issuance of the report and the attendant public discussion did seem to stop the growth in per capita cigarette consumption (graph).
While stopping the growth in smoking, it is worth considering what the tobacco companies were afraid would happen when the report came out.
Fortunately, we know, thanks to the millions of previously secret tobacco industry documents in the UCSF Legacy Tobacco Documents Library. In a word, the companies were terrified.
In June 1963, the major tobacco companies and their public relations agency, Hill and Knowlton, held discussions about the then-anticipated Surgeon General's report. Here is a summary of the meeting:
January 2, 2014
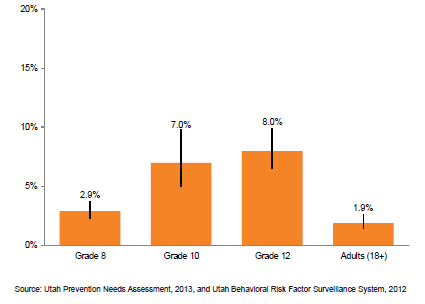 The Utah State Department of Health just published the results of their state survey on tobacco product use, which showed skyrocketing e-cigarette use among kids, reaching much higher levels than adults. Here are their conclusions:
The Utah State Department of Health just published the results of their state survey on tobacco product use, which showed skyrocketing e-cigarette use among kids, reaching much higher levels than adults. Here are their conclusions:
- The percentage of Utah students in • grades 8, 10, and 12 who reported that they had tried electronic cigarettes more than doubled from 2011 to 2013.
- Despite having no legal access to e-• cigarettes, Utah youth are three times more likely to report current use than adults.
- Nearly one third of Utah youth who • used e-cigarettes in the past 30 days report that they never tried conventional cigarettes.
- Due to candy-like flavors, aggressive • marketing, and lack of data regarding safety, monitoring the increasing use of e-cigarettes among youth is a public health priority.
The full report is available here.
December 29, 2013
Hadii Mamudu and I with several others just published "Multiple Streams Approach to Tobacco Control Policymaking in a Tobacco-Growing State" in Journal of Community Health. Here is the abstract:
Smokefree policies (SFPs) have diffused throughout theUS and worldwide. However, the development of SFPs in the difficult policy environment of tobacco-producing
states and economies worldwide has not been well explored. In 2007, Tennessee, the third largest tobacco producer in the US, enacted the Non-Smoker Protection Act
